Scientists are at present striving to retailer hydrogen extra effectively for a wide range of causes, and the most effective mediums is a kind of crystal known as perovskite. Chemical reactions can be utilized to interchange the oxygen ions within the crystalline powder with hydride (H-), turning it right into a perovskite oxyhydride. As soon as hydrogen is saved on this approach, it’s simply transportable and can be utilized as a catalyst to create ammonia. As ammonia is the principle ingredient in most fertilizers, is required for a lot of plastics, and is itself a kind of hydrogen gasoline, the perovskite oxyhydride energy has quite a few potential advantages. Nonetheless, whether or not they use excessive temperature or excessive stress, at present identified chemical reactions solely substitute about 17% of the oxygen with hydride, that means that the powder has the potential to retailer far more hydrogen than is at present doable.

Perovskite powder earlier than (left) and after (proper) conversion to perovskite oxyhydride.
The staff led by Chief Scientist Kobayashi is researching methods to extend the hydrogen saturation restrict from 17% and get extra hydrogen into perovskite powder. Somewhat than utilizing excessive temperatures or high-pressure strategies, they’ve been experimenting with bodily mechanochemical reactions, which work effectively at room temperature and make them a extra enticing possibility for sustaining the setting. Now, they’ve discovered a method to drastically enhance hydrogen saturation, with twice as many oxygen ions within the crystalline construction being changed by hydrides. Which means the brand new technique just about doubles the hydrogen-storage capability of perovskite powder.
Within the experiments, the researchers produced barium titanate oxyhydride in two methods: mechanochemically and topochemically. They discovered that the mechanochemical approach–bodily grinding and mixing the substances–had two benefits over the usual high-temperature technique. First, the lattice construction of the crystalline powder contained extra hydride. Second, even when taking items of every with the identical variety of hydrides, the mechanochemically produced model was a greater catalyst; extra ammonia was produced. Evaluation confirmed that this was as a result of the grinding course of induced useful deformations within the lattice that top warmth couldn’t.
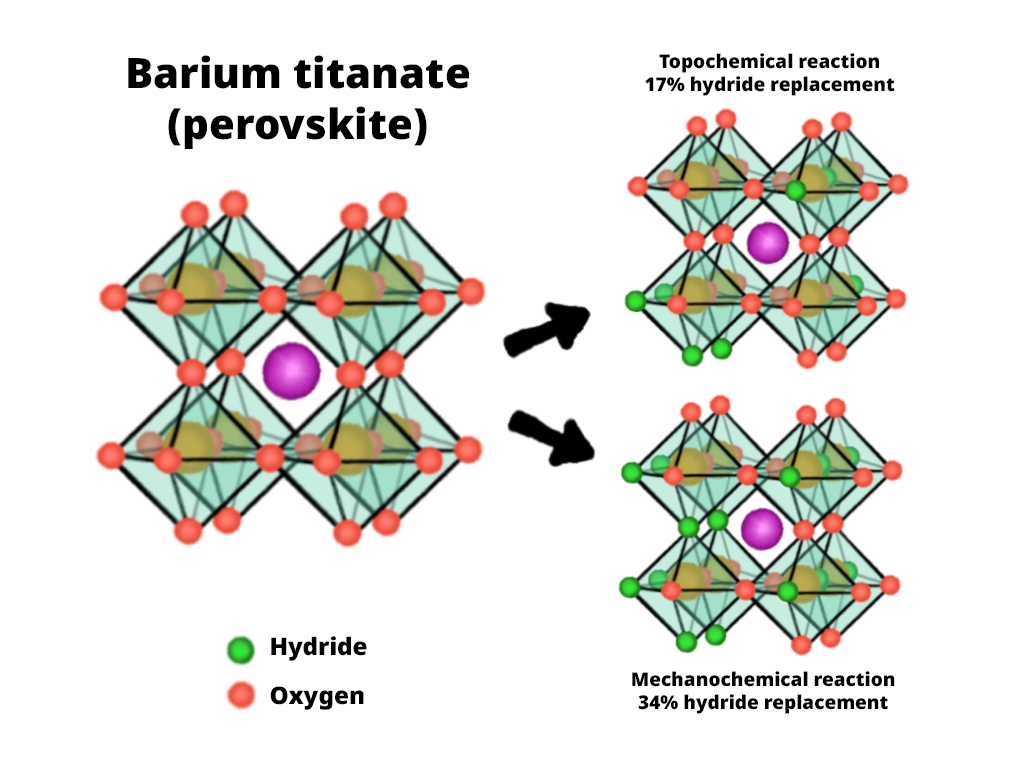
Barium titanate oxyhydride was produced from barium titanate through normal topochemical reactions (utilizing warmth) and the brand new mechanochemical reactions (by grinding). The mechanochemical response doubled the quantity of hydrogen that may very well be saved within the perovskite from 17% to 34%. As well as, the totally different construction allowed elevated ammonia manufacturing.
“This development is nice information for environmental sustainability and can ultimately assist us obtain an actual hydrogen-based economic system,” says Kobayashi. Within the brief time period, he says that their new findings present worthwhile materials design tips that will likely be helpful within the improvement of latest useful supplies that include hydride ions.
The brand new hydrogen saturation restrict of 34% is probably going the utmost that may be achieved utilizing barium titanate, however even higher outcomes could be doable beginning with one other perovskite. “In the long run,” Kobayashi says, “our mechanochemical method is predicted to yield even higher catalysts for ammonia synthesis, in addition to supplies for electrochemical gadgets resembling gasoline cells, a area wherein the Kobayashi Laboratory specializes.”
















.jpg)

